Lupin Manufacturing Solutions (NSE:LUPIN), a wholly owned subsidiary of Lupin Limited, has taken a strategic step forward with the expansion of its Dabhasa facility in India. This move underscores the company’s ambition to reinforce its position in the high-growth Contract Development and Manufacturing Organization (CRDMO/CDMO) space, particularly in complex therapeutics such as peptides.
Capacity Boost in Peptide Manufacturing
The expanded Dabhasa site now includes a new manufacturing block along with two specialized units dedicated to peptide production. This significantly enhances LMS’s ability to produce peptide building blocks, including protected amino acids—key components in modern drug development. The upgrade is designed to improve production flexibility while ensuring stringent global quality standards.
This expansion aligns with the broader pharmaceutical shift toward advanced modalities, where demand for peptide-based therapies and antibody-drug conjugates (ADCs) is rising rapidly. By scaling its capabilities, LMS is positioning itself as a key enabler for biotech and pharma innovators seeking reliable development and manufacturing partners.

Building a Global CRDMO Platform
LMS leverages the scientific expertise and regulatory experience of its parent company to offer end-to-end solutions—from early-stage development to commercial manufacturing. With a team of over 250 scientists and advanced infrastructure, the company is strengthening its integrated service offerings across complex chemistry and advanced modalities.
This strategic direction reflects Lupin’s broader vision of becoming a leading global CRDMO, supporting innovation across multiple therapeutic areas while expanding its global footprint.
Strong Financial Performance Supports Growth Strategy
Lupin Limited has reported a strong Q3 FY26 performance, highlighting the success of its growth strategy. Net sales reached ₹71,005 million, marking a 26.4% year-on-year increase, while EBITDA surged 61.8% to ₹22,095 million. Margins improved to 31.1%, reflecting operational efficiency and strong execution.
Net income stood at ₹11,756 million, demonstrating continued profitability despite some exceptional items. The company’s diversified revenue base across geographies played a key role in driving this growth.
Geographic and Segmental Strength
North America remains the largest revenue contributor, generating ₹32,183 million, followed by India at ₹20,387 million. The U.S. business recorded its highest-ever quarterly sales, supported by new launches in biosimilars and complex generics, including products like gRisperdal Consta® and Armlupeg.
Meanwhile, the India business grew 5.6% YoY, outperforming the market in key therapeutic segments such as cardiology, respiratory, and gastroenterology. Growth in emerging and developed international markets further reinforced Lupin’s global momentum.
Focus on Innovation and Future Growth
Lupin continues to invest heavily in R&D, manufacturing capabilities, and regulatory compliance to support its pipeline of complex generics, biosimilars, and specialty products. Multiple filings with the U.S. FDA highlight the company’s strong development pipeline.
Technical summary

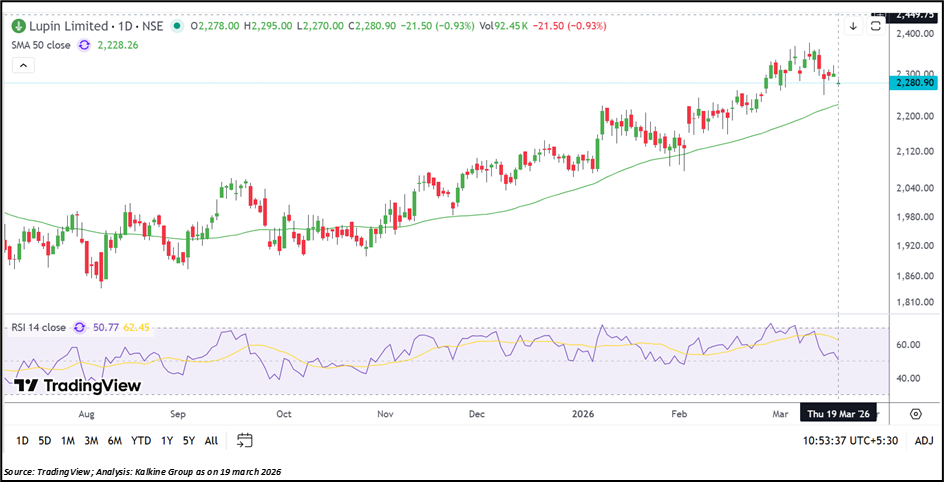
Lupin Limited is currently trading around ₹ 2,280.90, declining about 0.93% in the session, but continues to hold above its 50-day Simple Moving Average near ₹ 2,228.26, which remains an important trend indicator. The price structure reflects a steady uptrend with a pattern of higher highs and higher lows, indicating underlying strength despite recent consolidation.
Momentum has slightly cooled, with the 14-day RSI near 50.77, suggesting neutral conditions after the prior up move. Immediate support is seen around ₹ 2,100.00– ₹ 2,000.00, while ₹ 2,400.00– ₹ 2,550.00 may act as a resistance zone shaping the near-term price action.
Conclusion
The Dabhasa expansion is a strategic growth driver that strengthens Lupin’s positioning in the high-value CRDMO/CDMO space, particularly in peptides and complex therapeutics. By enhancing capacity and capabilities, the company is aligning with global demand for advanced drug development, while its strong financial performance and improving margins support sustained investments. Backed by a solid R&D pipeline, regulatory strength, and geographic diversification, Lupin is well-placed to capture long-term opportunities, improve scalability, and deliver consistent value through innovation-driven growth in the evolving pharmaceutical landscape.

